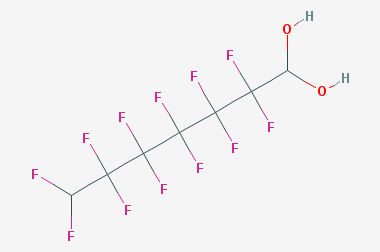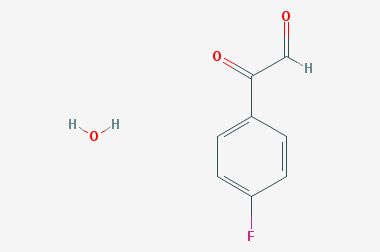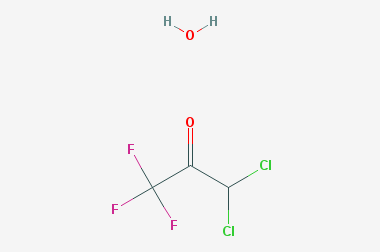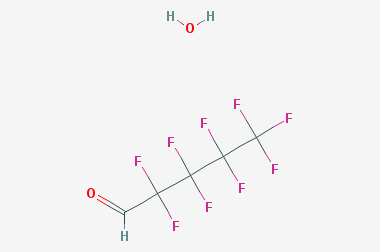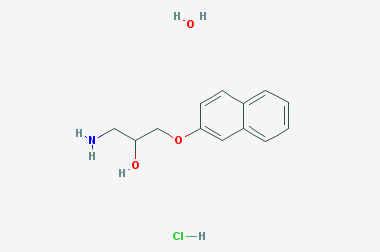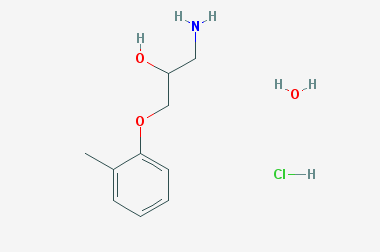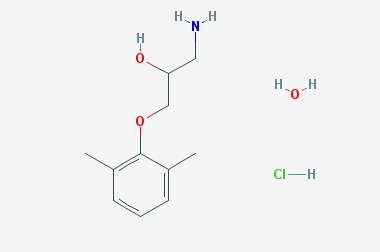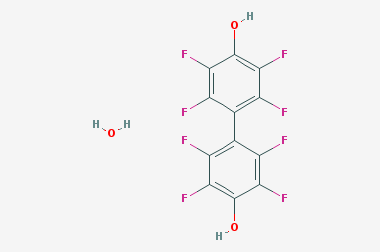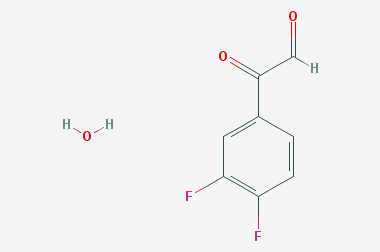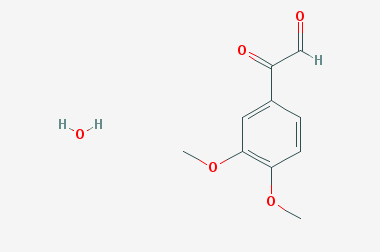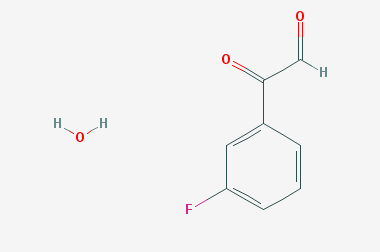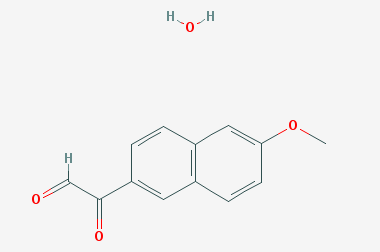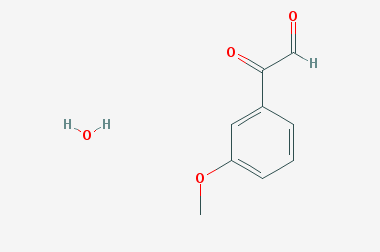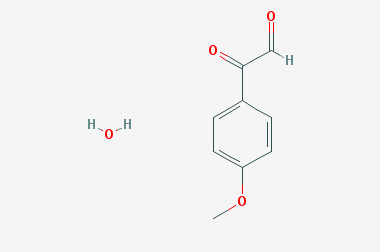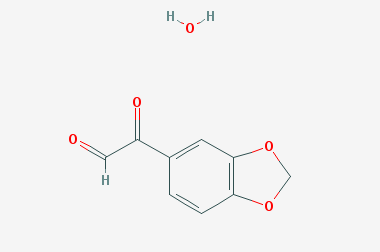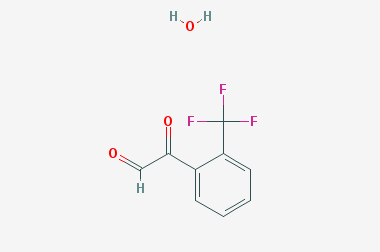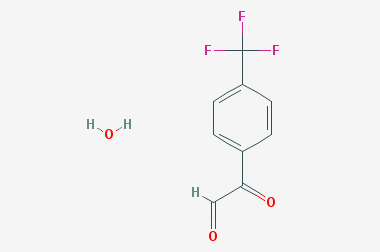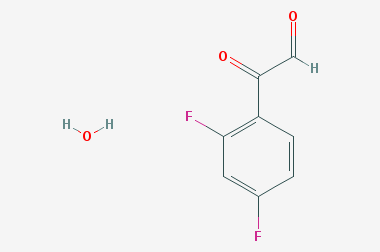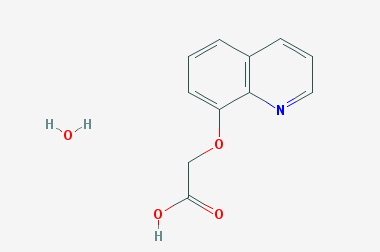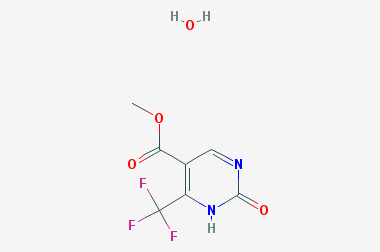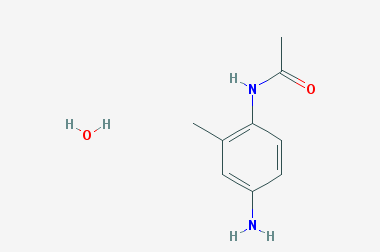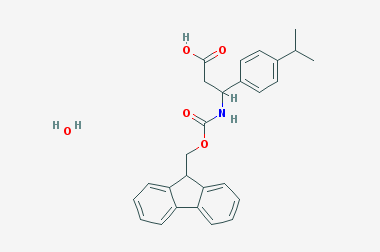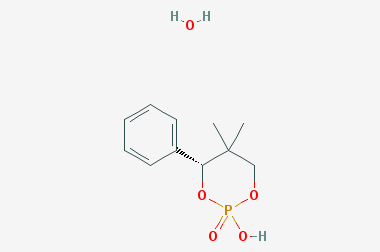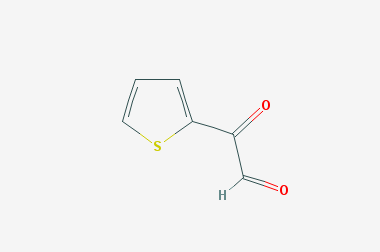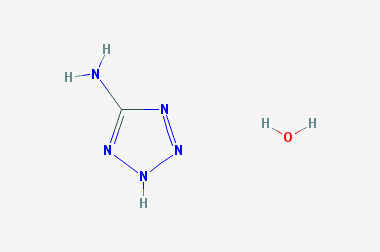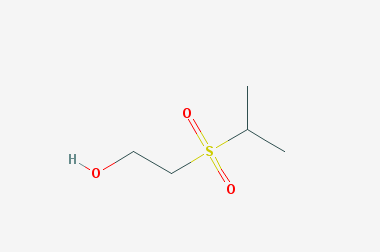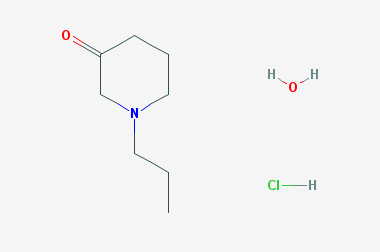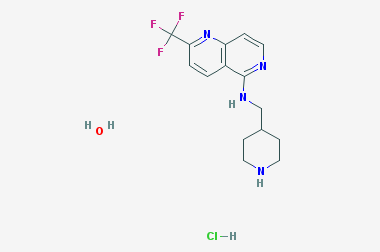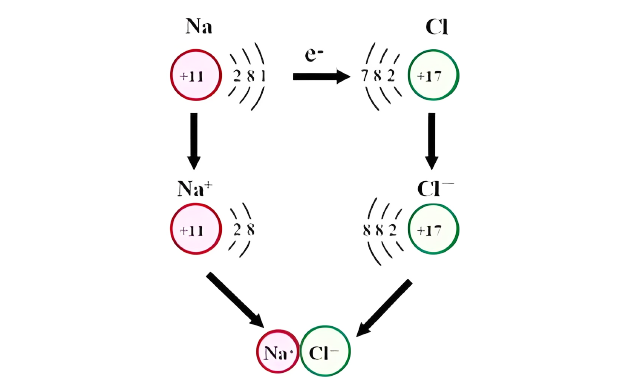
A reducing agent is a substance that loses electrons in a redox reaction. Generally speaking, a reactant in which the oxidation state of a certain substance increases is a reducing agent. Reducing agents themselves have reducing properties; when oxidized, the product is called an oxidation product. Reduction and oxidation reactions occur simultaneously; that is, while the reducing agent is undergoing a redox reaction with the reduced substance, it is also oxidized itself, becoming an oxidation product.
Selecting Products Filters
-
Product No.Product NameProduct Spec.BrandPrice